L-Tryptophan is an essential amino acid, meaning that it cannot be synthesized by the human body and must be obtained through dietary sources. It is a key building block for proteins and serves as a precursor for important molecules such as serotonin and melatonin. Here’s some information about its chemical structure and physical properties:
Chemical Structure of L-Tryptophan:
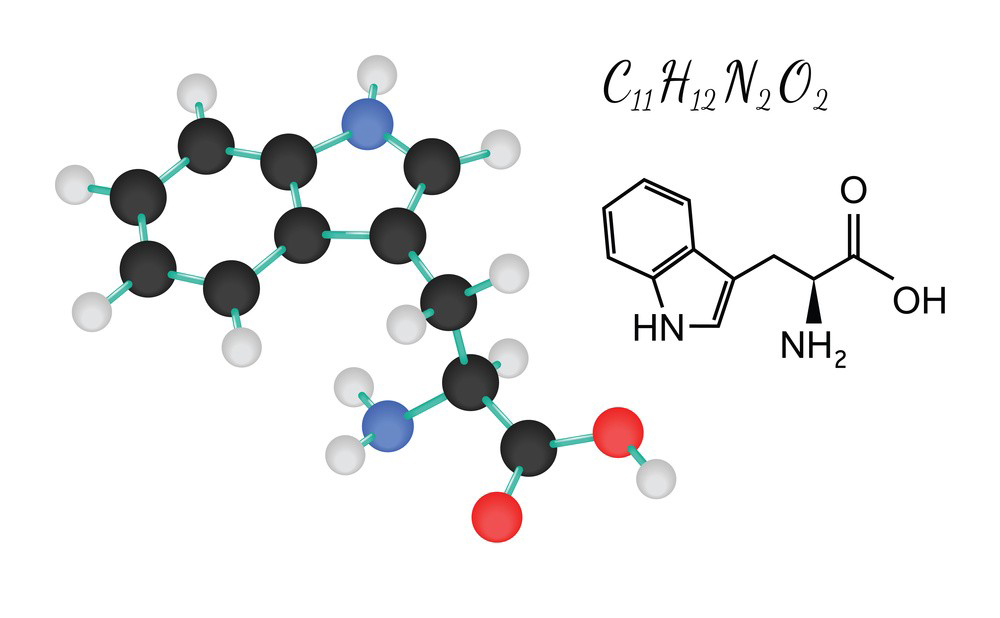
L-Tryptophan has the following chemical structure:
- Chemical Formula: C11H12N2O2
- IUPAC Name: (2S)-2-amino-3-(1H-indol-3-yl)propanoic acid
- Structural Formula:
H
|
H – C – COOH
|
NH2
- L-Tryptophan contains an indole ring, an amino group (-NH2), and a carboxyl group (-COOH) as part of its structure.
Physical Properties of L-Tryptophan:
The physical properties of L-Tryptophan include:
- Molecular Weight: The molecular weight of L-Tryptophan is approximately 204.23 grams per mole.
- Melting Point: L-Tryptophan has a melting point of about 295-314°C (563-597°F).
- Solubility: L-Tryptophan is sparingly soluble in water, meaning it dissolves poorly in water at room temperature. However, it is more soluble in hot water.
- Appearance: L-Tryptophan typically appears as a white to off-white, crystalline powder or solid.
- Odor and Taste: It is odorless and has a slightly bitter taste.
- Optical Activity: L-Tryptophan is chiral and exists in two enantiomeric forms, L-Tryptophan (levo-rotatory) and D-Tryptophan (dextro-rotatory). The L form is the one naturally occurring in proteins and is the biologically active form in humans.
- pKa Values: L-Tryptophan has two pKa values for its ionizable groups: pKa1 (carboxyl group) is approximately 2.38, and pKa2 (amino group) is approximately 9.39.
- UV Absorption: L-Tryptophan absorbs ultraviolet (UV) light at approximately 280 nanometers, which makes it useful in biochemical and biophysical studies.
- Stability: L-Tryptophan can be sensitive to light and air, and it may degrade over time if not stored properly. It is often recommended to store it in a cool, dry place away from direct light.
These physical properties are important in various scientific and medical applications, particularly in the fields of biochemistry, nutrition, and pharmaceuticals, where L-Tryptophan is used as a dietary supplement, a research tool, and a precursor for the synthesis of other biologically active compounds.
