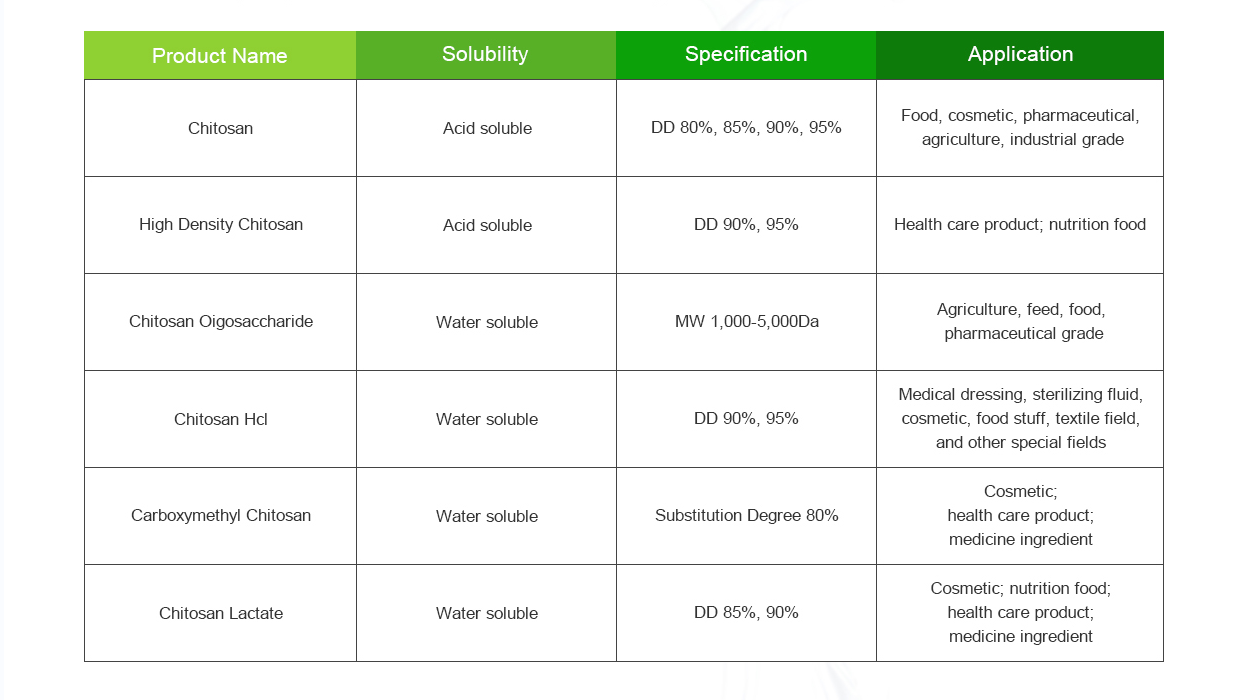
Chitosan is a biopolymer derived from chitin, which is found in the shells of crustaceans like shrimp and crabs. It has various applications in fields such as agriculture, food, pharmaceuticals, cosmetics, and biotechnology, due to its biodegradability, biocompatibility, and non-toxic nature. Here’s an overview of the materials and methods commonly associated with chitosan:
Materials of Chitosan:
- Chitosan: Chitosan is the main material of interest. It is obtained by deacetylating chitin, which is a natural polymer found in the shells of crustaceans.
- Chitin: The source material for chitosan. It can be extracted from crustacean shells through processes like deproteinization and demineralization.
- Solvents: Common solvents used for dissolving chitosan include acetic acid, hydrochloric acid, and citric acid.
- Crosslinking Agents: Substances like glutaraldehyde or tripolyphosphate are often used to crosslink chitosan, enhancing its stability and mechanical properties.
- Modifiers and Additives: Various modifiers and additives can be incorporated into chitosan formulations to tailor its properties, such as nanoparticles, proteins, and polymers.

Methods of Chitosan:
- Chitosan Preparation: Chitosan is typically prepared by deacetylating chitin using an alkali treatment. The resulting chitosan can be further processed and purified.
- Solution Preparation: Chitosan is dissolved in an appropriate solvent (e.g., acetic acid) to form a chitosan solution of desired concentration. This solution can be adjusted based on the intended application.
- Crosslinking: Crosslinking agents, such as glutaraldehyde, can be added to the chitosan solution to create a network structure that enhances its mechanical properties and stability.
- Film or Membrane Formation: Chitosan solutions can be cast into films or membranes using techniques like solvent casting, where the solvent is allowed to evaporate, leaving behind a chitosan film.
- Scaffold Fabrication: In tissue engineering, chitosan can be processed into three-dimensional scaffolds using methods like freeze-drying or electrospinning.
- Drug Delivery Systems: Chitosan can be used to encapsulate drugs or bioactive molecules, forming nanoparticles or microspheres for controlled release applications.
- Characterization: Various techniques can be employed to characterize chitosan-based materials, including Fourier-transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), X-ray diffraction (XRD), and mechanical testing.
- Biological and Biomedical Assays: Chitosan’s applications in wound healing, tissue engineering, and drug delivery often involve biological assays to evaluate its biocompatibility, antimicrobial properties, and efficacy.
It’s important to note that the specific materials and methods used with chitosan can vary widely based on the desired application. Researchers and scientists often modify and optimize these approaches to suit their particular needs and objectives.
The process of Chitosan
Chitosan is a biopolymer derived from chitin, which is a natural polymer found in the shells of crustaceans like shrimp, crabs, and lobsters, as well as in the cell walls of fungi. Chitosan has various applications, including in medicine, agriculture, food processing, and water treatment, due to its unique properties.
The process of obtaining chitosan involves several steps:
- Collection and Preparation of Raw Material: The primary source of chitosan is the shells of crustaceans. These shells are collected from seafood processing industries or other sources. The shells are usually cleaned to remove any remaining tissue, dirt, or impurities.
- Demineralization: The collected shells are typically rich in minerals like calcium carbonate. Demineralization involves treating the shells with an acidic solution, often hydrochloric acid, to dissolve and remove these minerals. This step helps in isolating chitin, the main precursor of chitosan.
- Deproteinization: After demineralization, the shells still contain proteins and other organic materials. Deproteinization is carried out by treating the shells with an alkaline solution, such as sodium hydroxide (NaOH), to remove proteins, lipids, and other impurities. This step results in the extraction of chitin.
- Deacetylation: Chitin extracted from the shells is composed of N-acetylglucosamine units. Chitosan is obtained by partially or completely deacetylating chitin through treatment with a strong alkali, such as NaOH, under controlled conditions. This process breaks some of the acetyl groups in chitin, resulting in the formation of chitosan.
- Washing and Filtration: After deacetylation, the chitosan is washed and filtered to remove any remaining chemicals, residues, or byproducts from the deacetylation process.
- Drying: The purified chitosan is then dried to remove excess moisture and obtain a stable powdered form that can be easily stored and used in various applications.
It’s important to note that the specific conditions and parameters of each step can vary depending on the desired properties of the chitosan and the intended application. The process outlined above is a general overview of how chitosan is typically produced, but variations and optimizations can be applied to suit different requirements.
Chitosan’s properties, such as biocompatibility, biodegradability, and ability to form films and gels, make it a versatile material with applications in areas such as wound healing, drug delivery, food preservation, and water purification.
