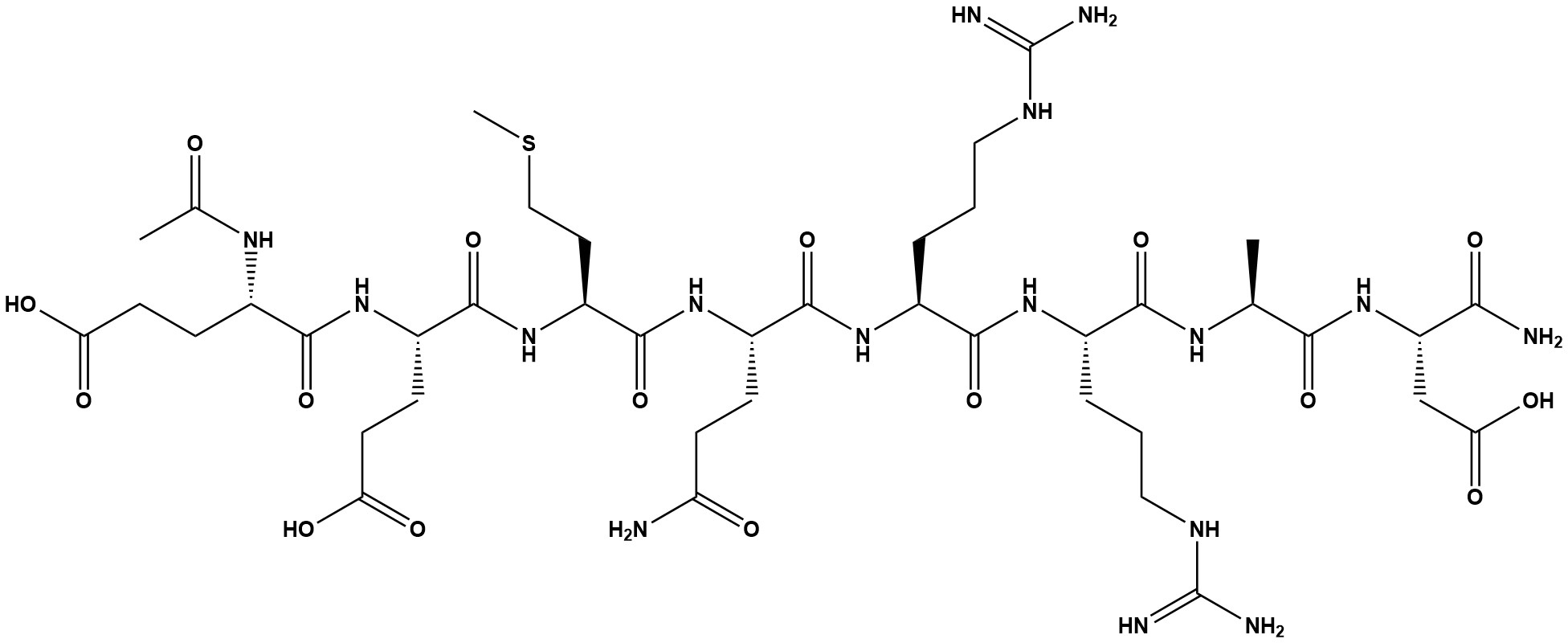
Acetyl Octapeptide-3/SNAP-8 is a synthetic peptide used in anti-wrinkle cosmetics. It is an octapeptide derived from the N-terminal fragment of the SNAP-25 protein and is produced through solid-phase peptide synthesis (SPPS) followed by purification and acetylation.
Below is a clear explanation of its synthesis process.
1. Raw Materials
The synthesis requires:
- Protected amino acids corresponding to the Acetyl Octapeptide-3/SNAP-8 sequence
- Solid resin support (commonly Wang resin or Rink amide resin)
- Coupling reagents (e.g., HBTU, HATU, or DIC)
- Deprotection reagent (typically piperidine)
- Cleavage reagent (usually trifluoroacetic acid, TFA)
The peptide sequence of Acetyl Octapeptide-3/SNAP-8 is derived from the N-terminal end of SNAP-25, a protein involved in neurotransmitter release.
2. Solid-Phase Peptide Synthesis (SPPS)
Step 1: Resin Loading
The C-terminal amino acid of the peptide is attached to a solid polymer resin.
This provides a stable anchor for sequential chain elongation.
Step 2: Deprotection
The Fmoc protecting group on the amino acid is removed using piperidine, exposing a free amino group.
Step 3: Amino Acid Coupling
The next Fmoc-protected amino acid is activated by coupling reagents and reacts with the free amine on the growing chain.
Reaction cycle:
- Deprotection
- Washing
- Coupling of the next amino acid
This cycle repeats until all eight amino acids are assembled.
3. N-Terminal Acetylation
After the peptide chain is completed:
- The N-terminus is acetylated using acetic anhydride or another acetylating agent.
- This forms Acetyl Octapeptide-3/SNAP-8, improving stability and bioactivity.

4. Cleavage from Resin
The peptide is cleaved from the solid support using:
- Trifluoroacetic acid (TFA) mixture
- Side-chain protecting groups are removed simultaneously.
This step releases the crude peptide.
5. Purification
The crude peptide is purified using:
- Reverse-phase High-Performance Liquid Chromatography (RP-HPLC)
Purification ensures:
- High purity (often >95%)
- Removal of truncated or incomplete peptides.
6. Characterization and Quality Control
The final peptide is confirmed using:
- Mass spectrometry (MS) – verifies molecular weight
- HPLC analysis – checks purity
- Amino acid analysis – confirms sequence composition
7. Final Product Processing
After purification:
- The peptide is lyophilized (freeze-dried) to obtain a stable powder.
- It is then dissolved in water or glycerin solutions for use in cosmetic formulations such as anti-wrinkle serums and creams.
Summary:
The synthesis of Acetyl Octapeptide-3/SNAP-8 involves solid-phase peptide synthesis, N-terminal acetylation, resin cleavage, HPLC purification, and analytical verification, producing a high-purity cosmetic peptide used for reducing expression wrinkles.
If you’d like, I can also explain:
- The exact amino-acid sequence of Acetyl Octapeptide-3/SNAP-8
- The chemical reaction mechanism during peptide coupling
- Or the industrial large-scale production process of Acetyl Octapeptide-3/SNAP-8.
