Ergothioneine is a naturally occurring, sulfur-containing derivative of the amino acid histidine that has attracted attention primarily for its antioxidant properties. Below is a detailed discussion of its chemical structure and physical properties.
Chemical Structure of Ergothioneine
Molecular Formula and IUPAC Name
- Molecular Formula: C₉H₁₅N₃O₂S
This relatively small molecule has a molecular weight of approximately 229.3 g/mol.
- IUPAC Name: One accepted systematic name is (2S)-3-[(1Z)-1-(3-carboxy-2-(trimethylazaniumyl)propyl)-2-thioxo-1,2-dihydro-3H-imidazol-3-yl]propanoic acid.
This lengthy IUPAC name reflects the molecule’s complexity, emphasizing its origin as a modified histidine.
Structural Features
1.Histidine Derivative:
- Ergothioneine is biosynthesized from histidine. Its side chain, originally an imidazole ring in histidine, is modified by the incorporation of sulfur. The molecule is often described as a “2-mercaptohistidine betaine” because of the replacement (or functionalization) of a hydrogen atom in the ring with a sulfur atom.
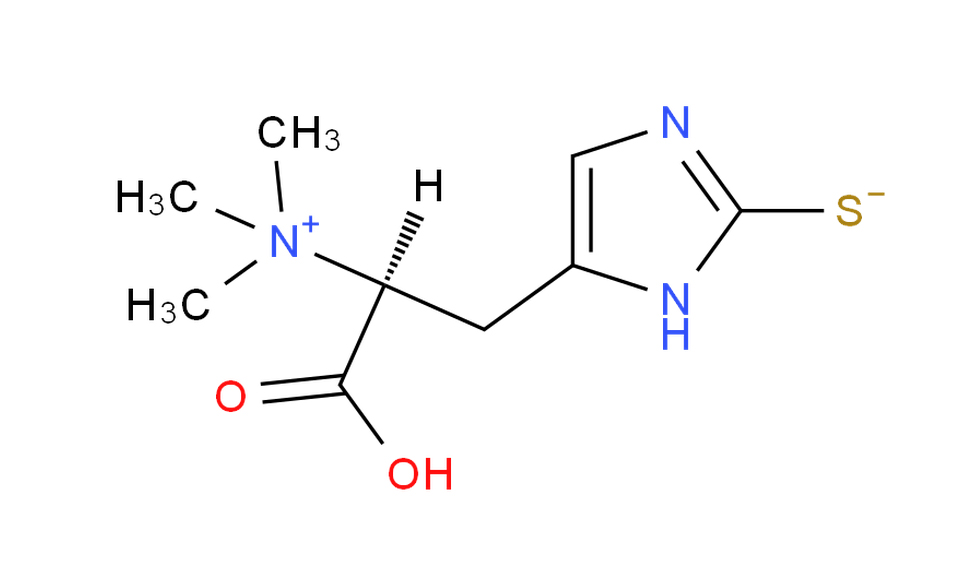
2.Thione/Thiol Tautomerism:
- One unique aspect of ergothioneine’s structure is its ability to exist in two tautomeric forms: the thiol form (–SH) and the thione form (–S=). Under physiological conditions, the thione tautomer predominates, a feature that contributes to its chemical stability and influences its antioxidant behavior.
3.Zwitterionic Character:
- Like many amino acid derivatives, ergothioneine exists as a zwitterion at physiological pH. This means that while it contains both positive (typically on the trimethylammonium group) and negative charges (from the carboxylate group), the overall molecule is electrically neutral.
4.Trimethylammonium Group:
- The presence of a trimethylammonium moiety (–N⁺(CH₃)₃) on the molecule not only contributes to its zwitterionic nature but also enhances its solubility in aqueous environments.
Physical Properties of Ergothioneine
Appearance and Solubility
1.Solid Form:
- Ergothioneine is typically isolated as a white crystalline powder. Its crystalline nature assists in its stability when stored under dry conditions.
2.Water Solubility:
- Due to its polar zwitterionic structure, ergothioneine is highly soluble in water. This high solubility is key to its biological role, as it circulates in bodily fluids and accumulates in tissues via a specific transporter.
3.Solvent Behavior:
- Although primarily water-soluble, the amphipathic character (bearing both hydrophilic and hydrophobic regions) may allow for limited solubility in polar organic solvents under appropriate conditions.
Thermal and Chemical Stability
1.Melting Point:
- While exact values can vary depending on crystallinity and purity, ergothioneine generally exhibits a moderate melting point characteristic of small organic crystalline compounds. Some literature sources report melting points in the vicinity of 140°C, though this can depend on the sample and conditions.
2.Stability:
- The thione tautomer’s dominance under physiological conditions contributes to the molecule’s stability against oxidation. This is in contrast to many other thiol-containing compounds (like glutathione) that are more prone to oxidation. Ergothioneine’s stability is a factor in its proposed protective effects against oxidative damage.
Acid–Base Properties
1.pKa Values:
As a zwitterionic molecule, Ergothioneine has multiple ionizable groups. Although the exact pKa values can vary slightly depending on the environment, general considerations include:
- A carboxyl group with a pKa typically in the acidic range (around 2–3),
- An amine (trimethylammonium) group with a pKa typically in the basic range (around 9–10).
The imidazole ring, modified by sulfur substitution, does not behave exactly like that of standard histidine but contributes to the overall distribution of charge across the molecule.
Redox and Antioxidant Behavior
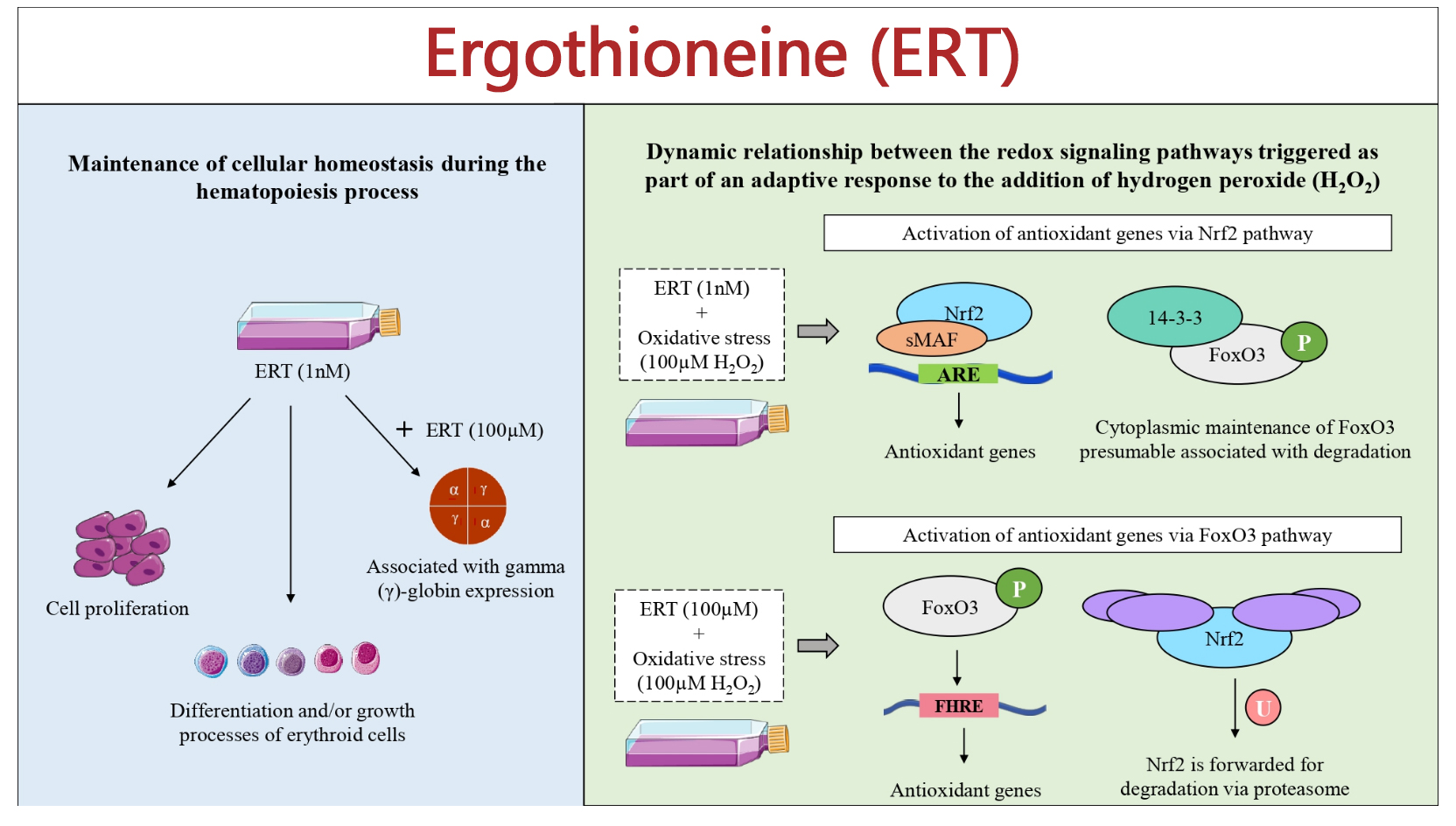
1.Antioxidant Function:
Ergothioneine’s antioxidant potential is partly a consequence of its ability to stabilize radical species via tautomerization and resonance within the imidazole ring. Its structure provides an effective trap for reactive oxygen species and other free radicals, thereby contributing to its cytoprotective effects in biological systems.

Summary
Ergothioneine is a small, water-soluble molecule with the formula C₉H₁₅N₃O₂S that features a modified histidine structure where the imidazole ring is functionalized with a sulfur atom. Predominantly existing in its thione form under physiological conditions, it displays zwitterionic characteristics and high stability—attributes that underpin its effectiveness as an antioxidant. Its physical properties, including its solubility in water and moderate melting point, make it well suited for its biological roles, including accumulation in cells through a specific transporter and protection against oxidative stress.
This comprehensive description should provide a solid understanding of both the chemical structure and the key physical properties of ergothioneine.
